“Can it heal?” Yes, tooth decay in the initial stages can heal if a cavity has not formed. In fact, even if you eventually need a filling, the measures you take to recover the tooth decay will stop the rapid progression of the decay and prevent the decay from reaching the canal and infecting it. Therefore, this topic should not be read only to avoid fillings. We can also try to cure decay to keep our teeth alive.
But how?
I will look at it from two perspectives:
1. What happens on the outer surface of the tooth:
According to the currently accepted theory of caries formation, the food we eat is broken down by the bacteria in our mouths, resulting in acid formation. This acid dissolves the minerals in the tooth enamel. This is called demineralization. Thanks to our saliva, this acid environment becomes neutral again, and the minerals in the saliva return to the tooth surface. This is called remineralization. This demineralization-remineralization cycle is a normal process, but if the minerals are frequently moving away from the tooth surface and cannot come back, that is, if the balance is disturbed, then caries will form in this area over time. So, the building mechanism cannot keep up with the rate of destruction.
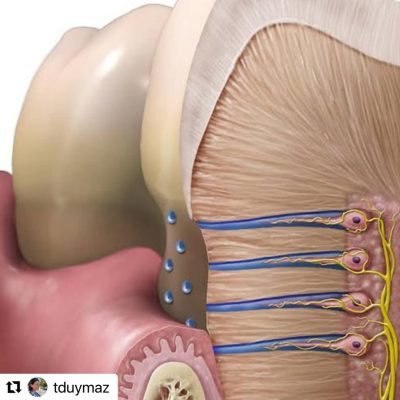
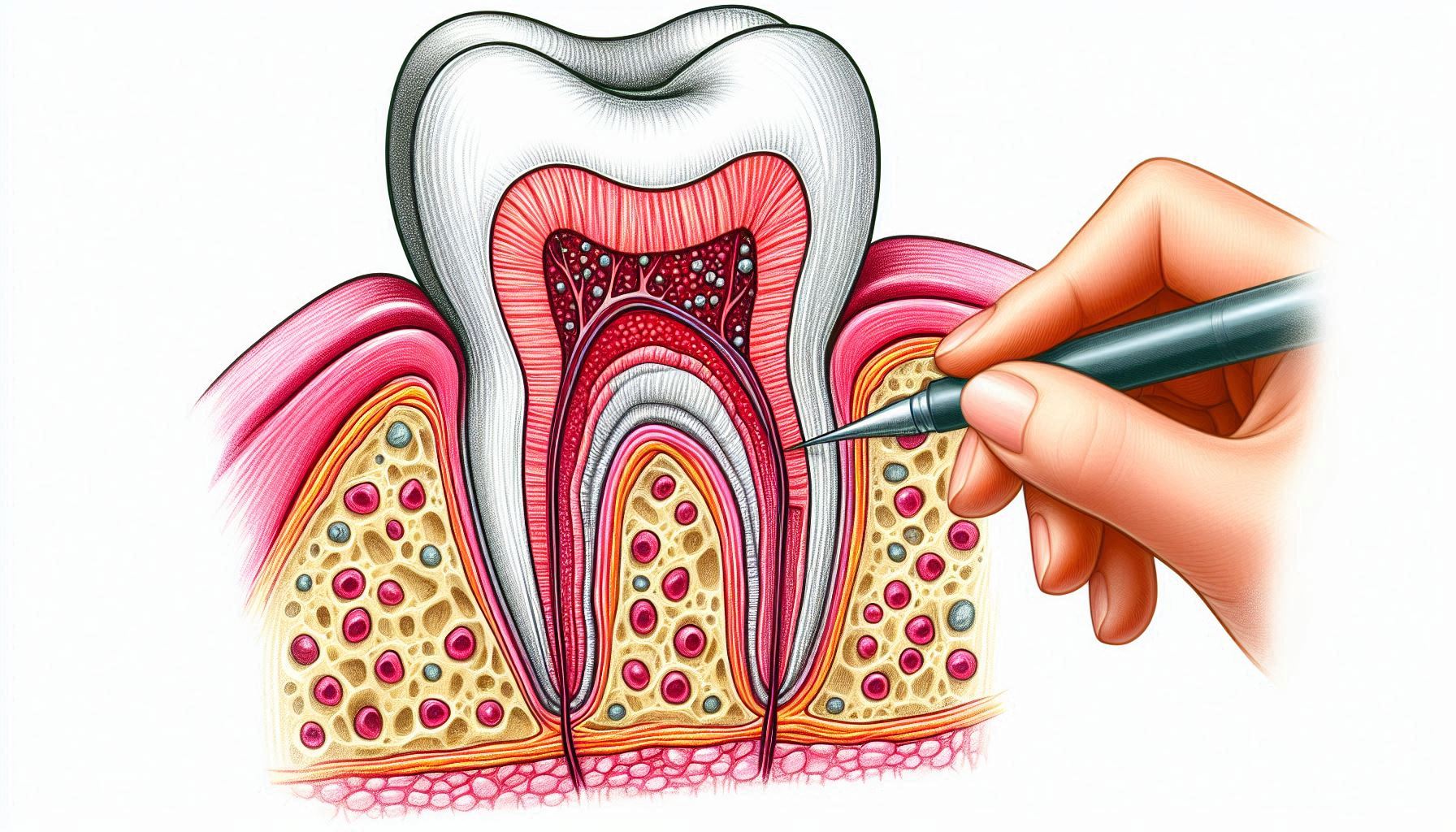
2. What happens inside the tooth:
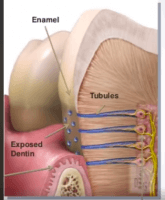
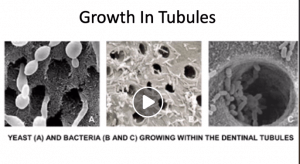
There is another repair mechanism that we dentists often encounter while filling, but for some reason, we do not emphasize stopping decay much. The repair that takes place inside the tooth, in the part we call “dentin.” Dentin is not just a mineral tissue. It contains canals and extensions of very special cells called odontoblasts. These sense threats from the outside and condense the dentin tissue to protect the tooth from the inside. There are also thousands of tiny canaliculi in the dentin, through which fluid flows from the inside of the tooth to the outside, which helps to keep bacteria out. Defence elements are also present here and actively protect the tooth.
In this case, we need to support our teeth externally and internally to protect them against cavities and heal incipient cavities. We can do the following.
Improve the quality of our saliva: There is a lot to say, but in short, saliva’s flow rate, pH, minerals, and immune system elements are important for healing caries. It is also important that our nutrition is not deficient, that we do not mouth breathe, and that we drink enough water. Vitamins A, D, and K2, minerals such as calcium and phosphorus, vitamin C, and adequate protein intake are important.
Oral flora: Acid-producing bacteria love sugar very much. When we consume it, it takes over in the mouth. As I wrote above, we need a diet that is free from packaged, processed foods and provides macro and micronutrients well.
Frequent snacking should be avoided because it doesn’t give the teeth time to remineralize. Especially if you eat junk food!
Avoid foods that stick to the teeth. These are usually processed foods. If you have to eat them, you can take measures such as rinsing your mouth or stimulating saliva flow by chewing gum after a meal.
Although there are different opinions about brushing your teeth immediately after a meal, it is not recommended. Brushing with paste can cause more mineral loss. You can wait half an hour.
To nourish the teeth from the inside: There are theories that vitamin K2 controls the flow of dentin fluid inside the teeth. Sugar reverses the direction of this flow, making the tooth unable to protect itself. Vitamin C can balance this. It is also necessary to provide the tooth with building blocks for repair. Calcium and fat-soluble vitamins come to mind again.